Planar
polymers of С60 undergo irreversible transformations
at high pressure, related with formation of new polymer bonds perpendicular to the
two-dimensional polymer planes. It is established that tetragonal (2D-T) polymer transforms into a new three-dimensional polymer phase at pressure ~20 GPa (Fig. 1).The high pressure phase in the
2D-T polymer is characterized
by narrow lines in
Raman spectrum and high degree of ordering. The new polymer phase of С60 is metastable
at normal pressure - upon heating to 580 К it transforms into a mixture of
monomer and dimer of С60. It is shown that the rhomboedric polymer (2D-R) transforms into a highly disordered phase at pressure ~14 GPa. The new phase is characterized by chaotic formation of polymer bonds
between molecules, belonging to neighboring planar polymer planes. This phase is also metastable at normal pressure and upon heating to
580 К it transforms into a mixture of
monomer and dimer of С60.
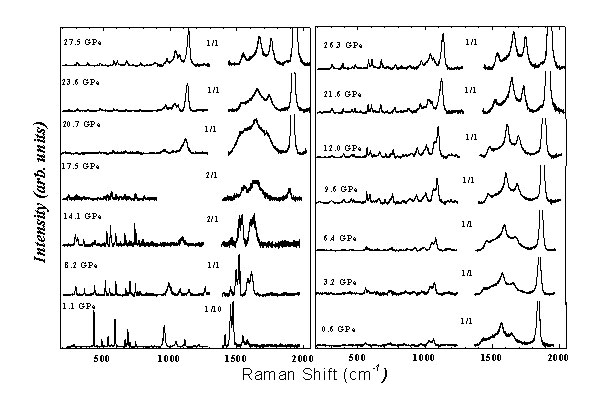
Fig. 1 Raman spectrum of a 2D-T polymer at pressure up to 27.5 GRa. Right graph - increase of pressure, left graph - decrease of pressure. At pressure about 20 GPa irreversible transition into a new high pressure phase occurs.